Quantification Assay of Diabetic Drug Sitagliptin in Pharmaceutical Preparations
A Review
DOI:
https://doi.org/10.5281/zenodo.15778061Abstract
 Abstract Views: 247
Abstract Views: 247
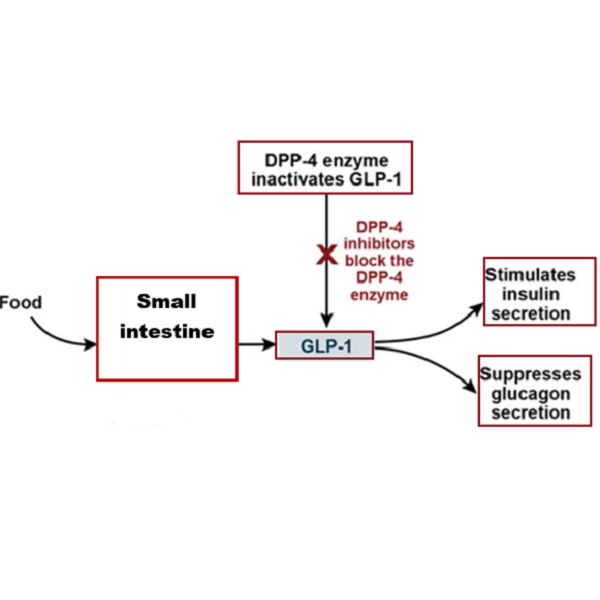
The diabetic drug Sitagliptin (STG) is a hypoglycemic drug of the dipeptidyl peptidase-4(DPP-4) inhibitor. The new therapeutic action by DPP-4 inhibitors depends on the decrease of the glucagon levels and stimulates glucose-dependent insulin release. This is done through inhibition of the inactivation of incretins, particularly glucagon-like peptide-1 (GLP-1) and gastric inhibitory polypeptide (GIP), thereby improving glycemic control. Quantification Assay of STG in pharmaceutical preparations is one of the researchers' interests because of its importance in the manufacturing of drugs, quality control of the dosage forms, and the accuracy in determining the dose. However, in biological fluids, determination of the drugs is of greater importance, as monitoring the drug, studying its pharmacokinetics, bioavailability, potency, the chemical and biological actions of the drugs and their metabolites in the body. This is a review of STG, its properties, action, and methods of determination.
Keywords:
Diabetic drug sitagliptin, Pharmaceutical preparations, Quantification assayReferences
Ahmed, R. M., Hadad, G. M., El-Gendy, A. E., & Ibrahim, A. (2018). Development of HPLC method for determination of sitagliptin in human plasma using fluorescence detector by experimental design approach. Analytical Chemistry Letters, 8(6), 813-828. https://doi.org/10.1080/22297928.2018.1545603
Anderson, P. O. (2018). Treating diabetes during breastfeeding. Breastfeeding Medicine, 13(4), 237-239. https://doi.org/10.1089/bfm.2018.0036
Bhargavi, P. D., Lolla, S., Sugunan, S., Gubbiyappa, K. S., Khan, A. A., Alanazi, A. M., & Nayak, B. V. (2023). The simultaneous quantification of Sitagliptin and Irbesartan in rat plasma using the validated LC-MS/MS method is applied to a pharmacokinetic study. Journal of Chromatography B, 1221, 123677. https://doi.org/10.1016/j.jchromb.2023.123677
de Paula e Mancilha, T., de Paula, C. E. R., Cassella, R. J., & Pacheco, W. F. (2013). Direct determination of sitagliptin in pharmaceutical formulations and its determination in urine after solid‐phase extraction by spectrofluorimetry. Luminescence, 28(6), 873-878. https://doi.org/10.1002/bio.2449
Disha, N. S., & Gurupadhayya, B. M. (2015). Spectrophotometric determination of Sitagliptin phosphate in bulk and pharmaceutical formulations. International Journal of Innovative Science, Engineering and Technology, 2(7), 702-709.
El-Bagary, R. I., Elkady, E. F., & Ayoub, B. M. (2011). Spectrophotometric methods for the determination of sitagliptin and vildagliptin in bulk and dosage forms. International Journal of Biomedical Science: IJBS, 7(1), 55. https://pubmed.ncbi.nlm.nih.gov/23675221/
Hirshberg, B., & Raz, I. (2011). Impact of the US Food and Drug Administration cardiovascular assessment requirements on the development of novel antidiabetes drugs. Diabetes Care, 34(Suppl 2), S101. https://doi.org/10.2337/dc11-s202
Nirogi, R., Kandikere, V., Mudigonda, K., Komarneni, P., Aleti, R., & Boggavarapu, R. (2008). Sensitive liquid chromatography tandem mass spectrometry method for the quantification of sitagliptin, a DPP‐4 inhibitor, in human plasma using liquid–liquid extraction. Biomedical Chromatography, 22(2), 214-222. https://doi.org/10.1002/bmc.926
Patil, S., Ramesh, B., Hareesh, A. R., Patil, K., & Dhokane, A. (2010). Development and Validation of RP-HPLC Method for the Estimation of Sitagliptin Phosphate in Tablet Dosage Form. Asian Journal of Research in Chemistry, 3(3), 653-655.
Rajeswari, A., Saritha, N., & Devanna, N. (2022). Validated RP-HPLC Method Development for Estimation of Ertugliflozin and Sitagliptin in Bulk and Dosage Forms. Journal of Pharmaceutical Research International, 34, 22-26.
Ravisankar, P., Mounika, G., Devadasu, C., & DevalaRao, G. (2014). A simple validated UV Spectrophotometric method for quantitative analysis of Sitagliptin phosphate in pharmaceutical dosage form. J. Chem. Pharmaceut. Sci, 7, 254.
Raza, A., Murtaza, S. H., Hanif, S., Iqbal, J., Ali, I., Aftab, T., ... & Syed, M. A. (2022). Validation of a rapid and economical RP-HPLC method for simultaneous determination of metformin hydrochloride and sitagliptin phosphate monohydrate: Greenness evaluation using AGREE score. Pakistan Journal of Pharmaceutical Sciences, 35(1).
Sekaran, C. B., & Rani, A. P. (2010). Development and validation of spectrophotometric method for the determination of DPP-4 inhibitor, sitagliptin, in its pharmaceutical preparations. Eclética Química, 35, 45-53. https://doi.org/10.1590/S0100-46702010000300003
Shantikumar, S., Sreekanth, G., SurendraNath, K. V., JaferValli, S., & Satheeshkumar, N. (2014). Compatibility study between sitagliptin and pharmaceutical excipients used in solid dosage forms. Journal of Thermal Analysis and Calorimetry, 115, 2423-2428. https://doi.org/10.1007/s10973-013-3329-3
Singh, D., Jain, N. K., Tiwari, A., & Khan, N. (2022). Development and Validation of Simultaneous Equation Method for Estimation of Sitagliptin and Saxagliptin in Combined Pharmaceutical Dosage Form by Using UV Spectrophotometric Method. International Journal of Medical Sciences and Pharma Research, 8(2), 83-90. https://doi.org/10.22270/ijmspr.v8i2.43
Sireesha, D., & Sravya, E. (2017). Development and validation of RP-HPLC method for the estimation of sitagliptin phosphate in tablet dosage form. International Journal of Applied Pharmaceutical Sciences and Research, 2(03), 41-45. https://doi.org/10.21477/ijapsr.v2i3.8099
Sirigiri, N., Subramanian, N. S., Reddy, G., & Kumar, N. (2018). Stability indicating method development and validation for simultaneous estimation of sitagliptin phosphate and metformin HCl in tablets by HPLC. International Journal of Pharmaceutical Sciences and Research, 9(10), 4294-4302.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Biomedicine and Chemical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.



















