Indirect Spectrophotometric Method for Determination of Methyldopa in Pure and Pharmaceutical Formulation
DOI:
https://doi.org/10.48112/bcs.v2i2.380Abstract
 Abstract Views: 991
Abstract Views: 991
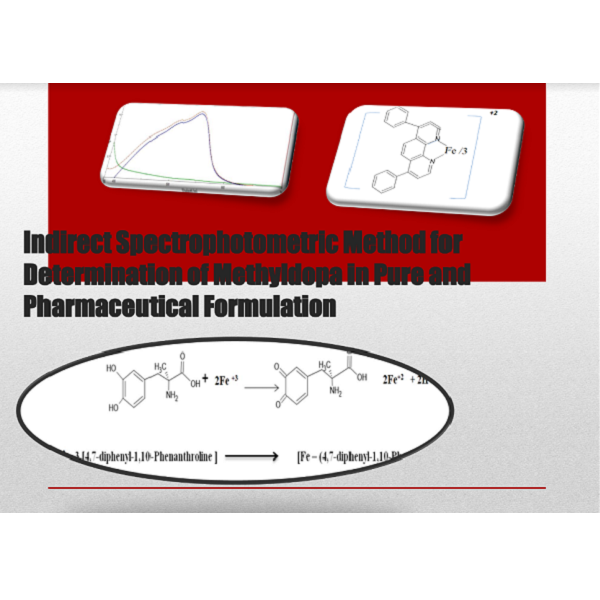
Sensitive and accurate spectrophotometric method for the assay of methyldopa (MeD) as pure form and in it is formulations. The method was centred on the oxidation of methyldopa drug with Fe+2ions. The free Fe+2 ions react with 4,7-diphenyl-1,10-phenanthroline chromogenic reagent. In the method, the formed Ferron complex is colorimetrically can be monitored at 533 nm versus reagent blank. The linear relationship between various MeD concentrations versus absorbance for each concentration was in the range of 0.25-2.5 μg.mL-1 with molar absorptivity 1.3272x105 L.mol-1.cm-1 for the method. The determination coefficient (R2) was found to be 0.9902, and the limit of detection (LOD) and limit of quantification (LOQ) were 0.011μg.ml-1and 0.036 μg.ml-1, respectively. The percent recovery from 99.84% to 102.40 %. The suggested procedure could be used for the estimation of MeD in dosage forms (two types of tablets from two companies) with satisfactory results.
Keywords:
Indirect spectrophotometric determination, methyldopa, 4,7-diphenyl-1, 10-phenanthroline chromogenic reagentReferences
Abdulrahman, L. K., Al-Abachi, A. M., & Al-Qaissy, M. H. (2005). Flow injection-spectrophotometeric determination of some catecholamine drugs in pharmaceutical preparations via oxidative coupling reaction with p-toluidine and sodium periodate. Analytica chimica acta, 538(1-2), 331-335. https://doi.org/10.1016/j.aca.2005.02.045
Abood, N. K., Hassan, M. J. M., & Al-Da’amy, M. A. (2019). Spectrophotometric determination methyldopa and salbutamol by oxidative coupling, cloud point and flow injection in pharmaceutical formulations. International Journal of Drug Delivery Technology, 9(2), 182-192.
AL-ghanam, D. N. A., & AL-Enizzi, M. S. S. (2022). Spectrophotometric determination of methyldopa by oxidative coupling reactions using 2, 4-dinitrophenylhydrazine reagent. Tikrit Journal of Pure Science, 27(5), 16-22. https://doi.org/10.25130/tjps.v27i5.10
Allah, N. M. G, Ahmed, A. M. K., & Tapabashi, N. O. (2022). Spectrophotometric Determination of Methyldopa in Pure and Pharmaceutical Preparations by the Oxidative Coupling Reaction with 1, 5-Diaminonaphthalene in the Presence of Ammonium Ceric (IV) Nitrate. Kirkuk University Journal-Scientific Studies, 17(4), 42-49. http://dx.doi.org/10.32894/kujss.2022.133098.1060
Antunes, R. S., Thomaz, D. V., Garcia, L. F., de Souza Gil, E., Sommerset, V. S., & Lopes, F. M. (2019). Determination of methyldopa and paracetamol in pharmaceutical samples by a low cost genipa americana l. polyphenol oxidase based biosensor. Advanced Pharmaceutical Bulletin, 9(3), 416. https://doi.org/10.15171%2Fapb.2019.049
da Silva Gonçalves, M., Armstrong, D. W., Cabral, L. M., Pinto, E. C., & de Sousa, V. P. (2021). Development and validation of a fast HPLC method for methyldopa enantiomers using superficially porous particle based macrocyclic glycopeptide stationary phase. Microchemical Journal, 164, 105957. https://doi.org/10.1016/j.microc.2021.105957
Dhamra, M.Y., & Al-Sabha, Th. N. (2020) Spectrophotometric method for indirect determination of antihypertensive drugs in pharmaceuticals. Egyptian Journal of Chemistry, 63(10), 3767-3777. https://doi.org/10.21608/ejchem.2020.18096.2102
Erdoğdu, G., Yağci, Ş. Z., & Savan, E. K. (2019). Investigation of the voltammetric behavior of methyldopa at a poly (p-Aminobenzene Sulfonic Acid) modified sensor. Turkish Journal of Pharmaceutical Sciences, 16(4), 450. https://doi.org/10.4274%2Ftjps.galenos.2018.44711
Gadkariem, E. A., Ibrahim, K. E. E., Kamil, N. A. A., Haga, M. E. M., & El-Obeid, H. A. (2009). A new spectrophotometric method for the determination of methyldopa. Saudi pharmaceutical journal, 17(4), 289-293. https://doi.org/10.1016/j.jsps.2009.10.005
Kim, B. K., & Koda, R. T. (1977). Fluorometric determination of methyldopa in biological fluids. Journal of pharmaceutical sciences, 66(11), 1632-1634. https://doi.org/10.1002/jps.2600661135
Nabeel, S. O, Mohanad, M. S., & Shielan A. O.(2017). Indirect spectrophotometric methods in determination of cefadroxil in pharmaceutical preparations, Raf.J.Sci., 26(1): 56-65. http://dx.doi.org/10.33899/rjs.2017.139095
Nejres, A. M., & Najem, M. A. (2023). A Novel Yttrium (III) Complex for Estimating Dopamine in Pure and Pharmaceutical Dosage Forms. Biomedicine and Chemical Sciences, 2(1), 23-30. https://doi.org/10.48112/bcs.v2i1.323
Ribeiro, P. R. S., Pezza, L., & Pezza, H. R. (2005). Spectrophotometric determination of methyldopa in pharmaceutical formulations. Ecletica Quimica, 30, 23-28.
Sanati, A. L., & Faridbod, F. (2017). Electrochemical determination of methyldopa by graphene quantum dot/1-butyl-3-methylimidazolium hexafluoro phosphate nanocomposite electrode. Int. J. Electrochem. Sci, 12(9), 7997-8005.
Sweetman, S. C. (2009). Martindale: the complete drug reference. Pharmaceutical press.
Yanik, S., Betül, A. R. İ., Demirci, S., & Yilmaz, S. (2020). Development of electrochemical sensors for quantitative analysis of methyldopa at modified-GCE and PGE electrodes by voltammetry. Journal of Scientific Perspectives, 4(4), 223-236.
Zečević, M., Živanović, L. J., Agatonovic-Kustrin, S., & Minic, D. (2001). The use of a response surface methodology on HPLC analysis of methyldopa, amiloride and hydrochlorothiazide in tablets. Journal of pharmaceutical and biomedical analysis, 24(5-6), 1019-1025. https://doi.org/10.1016/S0731-7085(00)00536-7
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Biomedicine and Chemical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.



















