In Silico Study, Synthesis and Evaluation of Cytotoxic Activity of New Sulfonamide-Isatin Derivatives as Carbonic Anhydrase Enzyme Inhibitors
DOI:
https://doi.org/10.5281/zenodo.15777417Abstract
 Abstract Views: 425
Abstract Views: 425
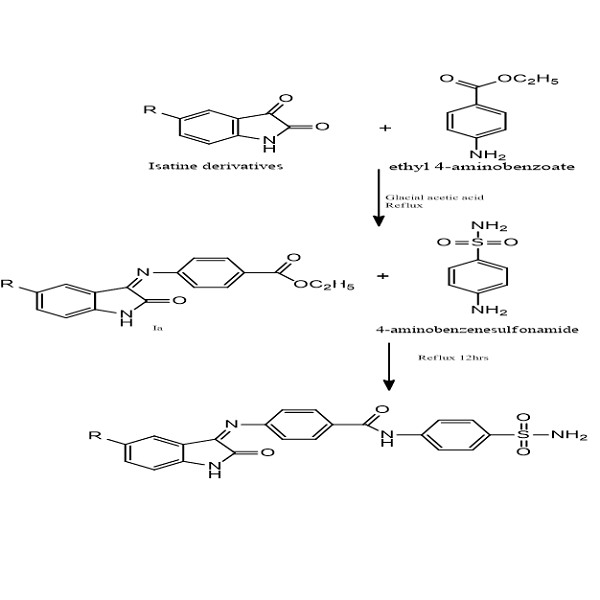
Design, molecular docking, synthesis and evaluation of cytotoxic activity of new compounds I, II, III and IV that have isatin-sulfonamide derivatives. For chemical synthesis, chemical compounds such as sulfonamide, 4-aminoethyl benzoate, isatin and its derivatives were used. For the docking study, the MOE software program version 2015.10 was used. And MTT assay for the prediction of cytotoxic activity. The synthesised compounds demonstrated significant inhibition of carbonic anhydrase XII activity through molecular docking and significant inhibition of cancer cell viability. Compounds II and IV show higher S-scores than acetazolamide. Also, the MTT assay shows IC50 against MCF-7 cells (0.06 µM and 0.105 µM) of compounds II and IV, respectively, when compared with IC50 0.394 µM of acetazolamide. IC50 against Hct116 cells (0.063 µM and 0.114 µM) of compounds II and IV, respectively, when compared with IC50 0.901 µM of acetazolamide. The MTT assay explains that compounds II and IV have better cytotoxic activity compared with acetazolamide. New compounds that were produced showed signs of cytotoxicity and carbonic anhydrase inhibitory qualities.
Keywords:
Carbonic anhydrase, Docking study, SulfonamideReferences
Abbas, Z. K., Naser, N. H., & Atiya, R. N. (2023). Targeting the Carbonic Anhydrase Enzyme with Synthesized Benzenesulfonamide Derivatives: Inhibiting Tumor Growth. Journal of Contemporary Medical Sciences, 9(4).
Aggarwal, M. (2013). Insights into human carbonic anhydrase inhibitor design (Doctoral dissertation, University of Florida).
Alibeg, A. A. A., & Mohammed, M. H. (2024). Design, synthesis, insilco study and biological evaluation of new isatin-sulfonamide derivatives by using mono amide linker as possible as histone deacetylase inhibitors. Polski Merkuriusz Lekarski: Organ Polskiego Towarzystwa Lekarskiego, 52(2), 178-188. https://doi.org/10.36740/merkur202402106
Alibeg, A. A. A., & Mohammed, M. H. (2024). Molecular docking, synthesis, characteristics and preliminary cytotoxic study of new coumarin-sulfonamide derivatives as histone deacetylase inhibitors. Wiadomosci lekarskie (Warsaw, Poland: 1960), 77(3), 514-525.
Aljubouri, R. S., & Naser, N. H. (2023). In Silico Study of New Carbonic Anhydrase Inhibitor Derivatives Bearing 1, 3, 4-Oxadiazole Moiety with Promising Anti-Cancer Activity. Journal of Contemporary Medical Sciences, 9(5).
Al-Khuzaie, M. G., Fahad, M. M., & Al-Safi, A. J. (2022). Synthesis, Reaction and Biological Importance of Isatin Derivatives. Biomedicine and Chemical Sciences, 1(3), 193-206.
Altalhi, A. A. (2024). Novel N-heterocyclic Schiff base based on Isatin derivative as a sustainable, eco-friendly, and highly efficiency corrosion inhibitor for carbon steel in sulfuric acid medium: Electrochemical and Computational investigation. International Journal of Electrochemical Science, 19(1), 100449. https://doi.org/10.1016/j.ijoes.2023.100449
Aziz Alibeg, A. A., Abdulsada, A. H., & Nasser, N. H. (2020). Design and Synthesis of Possible Mutual Prodrugs of (Nsaid) Etodolac and Tolmetin with (Cytotoxic) Gemcitabine. Systematic Reviews in Pharmacy, 11(11), 315-318.
Dorak, M. T., & Karpuzoglu, E. (2012). Gender differences in cancer susceptibility: an inadequately addressed issue. Frontiers in Genetics, 3, 268. https://doi.org/10.3389/fgene.2012.00268
Edgren, G., Liang, L., Adami, H. O., & Chang, E. T. (2012). Enigmatic sex disparities in cancer incidence. European Journal of Epidemiology, 27, 187-196. https://doi.org/10.1007/s10654-011-9647-5
Machlowska, J., Baj, J., Sitarz, M., Maciejewski, R., & Sitarz, R. (2020). Gastric cancer: epidemiology, risk factors, classification, genomic characteristics and treatment strategies. International Journal of Molecular Sciences, 21(11), 4012. https://doi.org/10.3390/ijms21114012
Naji, E. M., Naser, N. H., & Hussein, S. A. (2023). In silico study, synthesis, and antineoplastic evaluation of thiazole-based sulfonamide derivatives and their silver complexes with expected carbonic anhydrase inhibitory activity. Journal of Medicine and Life, 16(12), 1857. https://doi.org/10.25122/jml-2023-0180
Noor, H. N. (2017). β-Carbonic Anhydrase as a Target for Eradication of Mycobacterium tuberculosis. OAJPR, 1(1), 000106.
Patil, J. V., Umar, S., Soni, R., Soman, S. S., & Balakrishnan, S. (2023). Design, synthesis and anticancer activity of amide derivatives of substituted 3-methyl-benzofuran-2-carboxylic acid. Synthetic Communications, 53(3), 217-233. https://doi.org/10.1080/00397911.2022.2160648
Pavia, D. L., Lampman, G. M., Kriz, G. S., & Vyvyan, J. A. (2014). Introduction to Spectroscopy. Nelson Education.
Sağlık, B. N., Cevik, U. A., Osmaniye, D., Levent, S., Çavuşoğlu, B. K., Demir, Y., ... & Kaplancıklı, Z. A. (2019). Synthesis, molecular docking analysis and carbonic anhydrase I-II inhibitory evaluation of new sulfonamide derivatives. Bioorganic chemistry, 91, 103153. https://doi.org/10.1016/j.bioorg.2019.103153
Salerno, S., Amendola, G., Angeli, A., Baglini, E., Barresi, E., Marini, A. M., ... & Taliani, S. (2021). Inhibition studies on carbonic anhydrase isoforms I, II, IV and IX with N-arylsubstituted secondary sulfonamides featuring a bicyclic tetrahydroindazole scaffold. European Journal of Medicinal Chemistry, 220, 113490. https://doi.org/10.1016/j.ejmech.2021.113490
Shaldam, M. A., Almahli, H., Angeli, A., Badi, R. M., Khaleel, E. F., Zain-Alabdeen, A. I., ... & Tawfik, H. O. (2023). Discovery of sulfonamide-tethered isatin derivatives as novel anticancer agents and VEGFR-2 inhibitors. Journal of Enzyme Inhibition and Medicinal Chemistry, 38(1), 2203389. https://doi.org/10.1080/14756366.2023.2203389
Siegel, R. L., Miller, K. D., & Jemal, A. (2019). Cancer statistics, 2019. CA: A Cancer Journal for Clinicians, 69(1), 7-34. https://doi.org/10.3322/caac.21551
Tafreshi, N. K., Lloyd, M. C., Bui, M. M., Gillies, R. J., & Morse, D. L. (2014). Carbonic anhydrase IX as an imaging and therapeutic target for tumors and metastases. Carbonic Anhydrase: Mechanism, Regulation, Links to Disease, and Industrial Applications, 221-254. https://doi.org/10.1007/978-94-007-7359-2_12
Tuğrak, M., Gül, H. İ., Sakagami, H., Kaya, R., & Gülçin, İ. (2021). Synthesis and biological evaluation of new pyrazolebenzene-sulphonamides as potentialanticancer agents and hCA I and II inhibitors. Turkish Journal of Chemistry, 45(3), 528-539. https://doi.org/10.3906/kim-2009-37
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Biomedicine and Chemical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.



















