Synthesis, Characterization and Theoretical Study of 2,4-diaryl-1,3-Selenazole
DOI:
https://doi.org/10.5281/zenodo.15772101Abstract
 Abstract Views: 360
Abstract Views: 360
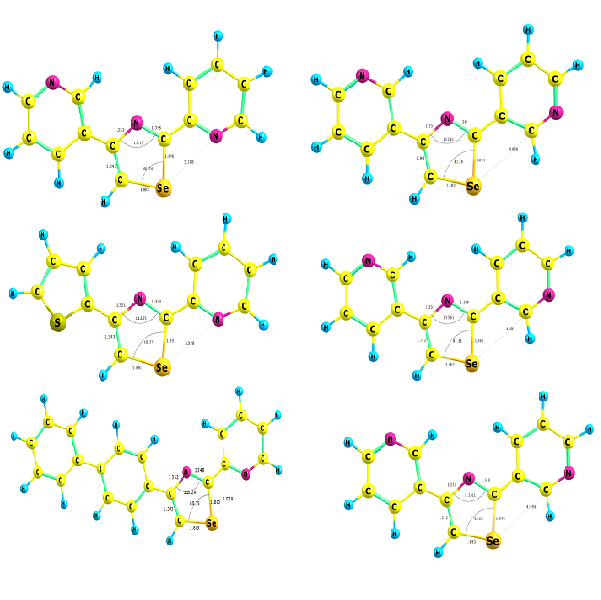
The synthesis of two new 2,4-diaryl-1,3-selenazoles series were achieved by subjecting primary arylselenocarboxylic amides to cyclization with α-bromoketones. The systematic preparation of 2,4-diaryl-1,3-selenazoles was carried out by the primary selenoamides with α-halo ketones. Selenoamides were obtained from the reaction of NaHSe with aryl nitrile .The resulting 2,4-diary-1,3-selenazoles have been characterized by different spectroscopic methods; such as 1H ,13C NMR, IR, and mass spectroscopic data to prove the structural formula of the prepared compounds. Theoretical study has been carried out by using DFT theory to study the structural and electronic properties of prepared compounds.
Keywords:
2,4-diaryl-1,3-selenazoles, NaHSe, organoselenium compounds, α-bromoketonesReferences
Abdullah, S. A., Ali, D. S., & Hameed, A. J. (2020). A study of the biological activity of 4-(Para-Substituted phenyl)-1, 2, 3-Selenadiazole derivatives as anti-oxidant and anti-breast cancer. Basrah Journal of Science, 38(3), 480-496.
Alias, N. Z., Idris, M. H. M., Somat, N. A., Mahmud, N., Hasan, S., Lam, K. W., ... & Hassan, N. I. (2020). Synthesis, antioxidant, anticholinesterase activities and molecular docking studies of coumaryl 1, 3-selenazoles derivatives. Preprints.
Al-Rubaie, A. Z., Al-Masoudi, W. A., Hameed, A. J., Yousif, L. Z., & Graia, M. (2008). Synthesis, reaction and antiviral activity of 2, 4-diaryl-1, 3-selenazoles. Journal of the Korean Chemical Society, 52(1), 36-46.
Al-Rubaie, A. Z., Yousif, L. Z., & Al-Hamad, A. J. (2002). Palladium-catalyzed formation of 3, 5-diaryl-1, 2, 4-selenadiazoles from arylselenocarboxamide. Journal of Organometallic Chemistry, 656(1-2), 274-280. https://doi.org/10.1016/S0022-328X(02)01631-5
Alsawad, D. H., Al-Riyahee, A. A., & Hameed, A. J. (2021, November). Simulation Study of Structural and Electronic Properties for Adducts complexes of Bis (Acetylacetonato) oxoVandium (IV) with 4-(Para-substituted phenyl)-1, 2, 3-Selenadiazole. In Journal of Physics: Conference Series (Vol. 2063, No. 1, p. 012002). IOP Publishing. https://doi.org/10.1088/1742-6596/2063/1/012002
Atkins, P. (2010). Shriver and Atkins' inorganic chemistry. Oxford University Press, USA.
Beswick, M. A., Harmer, C. N., Raithby, P. R., Steiner, A., Tombul, M., & Wright, D. S. (1999). Reactions of organolithium complexes with elemental selenium: insertion producing [PhC CSeLi· TMEDA· THF] 1 and cyclisation producing [{PhC} 4Se] 2 (TMEDA=[Me2NCH2] 2). Journal of organometallic chemistry, 573(1-2), 267-271. https://doi.org/10.1016/S0022-328X(98)00830-4
Cordes, D. B., Hua, G., Slawin, A. M., & Woollins, J. D. (2011). 2, 5-Diaryl-1, 3, 4-selenadiazoles prepared from Woollins' reagent. Crystal Structure Communications, 67(12), o509-o514. https://doi.org/10.1107/S0108270111049900
Dzedulionytė, K., Voznikaitė, P., Bieliauskas, A., Malinauskienė, V., Sløk, F. A., & Šačkus, A. (2021). Methyl 2-amino-4-[1-(tert-butoxycarbonyl) azetidin-3-yl]-1, 3-selenazole-5-carboxylate. Molbank, 2021(2), M1207. https://doi.org/10.3390/M1207
Etesam, L., Mosslemin, M. H., & Hassanabadi, A. (2022). A simple, one-pot method for the synthesis of functionalized selenazole derivatives. Phosphorus, Sulfur, and Silicon and the Related Elements, 197(12), 1273-1276. https://doi.org/10.1080/10426507.2022.2096609
Geisler, K., Jacobs, A., Künzler, A., Mathes, M., Girrleit, I., Zimmermann, B., ... & Langer, P. (2002). Efficient synthesis of primary selenocarboxylic amides by reaction of nitriles with phosphorous (V) selenide. Synlett, 2002(12), 1983-1986. https://doi.org/10.1055/s-2002-35604
Hansch, C., & Leo, A. (1995). Exploring quantitative structure-activity relationships (QSAR); Fundamentals and applications in chemistry and biology. American Chemical Society.
Hua, G., Cordes, D. B., Du, J., Slawin, A. M., & Woollins, J. D. (2018). Diverse derivatives of selenoureas: A synthetic and single crystal structural study. Molecules, 23(9), 2143. https://doi.org/10.3390/molecules23092143
Hua, G., Du, J., Slawin, A. M., & Woollins, J. D. (2014). A synthetic and structural study of arylselenoamides and 2, 4-diaryl-1, 3-selenazoles. Synlett, 25(15), 2189-2195. https://doi.org/10.1055/s-0034-1378525
Hua, G., Henry, J. B., Li, Y., Mount, A. R., Slawin, A. M., & Woollins, J. D. (2010). Synthesis of novel 2, 5-diarylselenophenes from selenation of 1, 4-diarylbutane-1, 4-diones or methanol/arylacetylenes. Organic & Biomolecular Chemistry, 8(7), 1655-1660. https://doi.org/10.1039/B924986A
Hua, G., Li, Y., Fuller, A., Slawin, A. M. Z., & Woollins, J. D. (2009). Facile synthesis and structure of novel 2, 5-disubstituted 1, 3, 4-selenadiazoles. European Journal of Organic Chemistry, 2009(10), 1612-1618. https://doi.org/10.1002/ejoc.200900013
Jalbout, A. F., Hameed, A. J., & Essa, A. H. (2008). Structural isomers of 2-(2, 3 and 4-substituted-phenyl)-1, 2-benzisoselenazol-3 (2H)-one: A Theoretical Study. Journal of Organometallic Chemistry, 693(12), 2074-2078. https://doi.org/10.1016/j.jorganchem.2008.02.026
Jalbout, A. F., Hameed, A. J., Jimenez-Fabian, I., Ibrahim, M., & De Leon, A. (2008). Chalcanthrene–fullerene complexes: A theoretical study. Journal of Organometallic Chemistry, 693(2), 216-220. https://doi.org/10.1016/j.jorganchem.2007.09.036
Koketsu, M., & Ishihara, H. (2003). Synthesis of 1, 3-selenazine and 1, 3-selenazole and their biological activities. Current Organic Chemistry, 7(2), 175-185. https://doi.org/10.2174/1385272033373102
Li, Y., Hua, G. X., Slawin, A. M., & Woollins, J. D. (2009). The X-ray crystal structures of primary aryl substituted selenoamides. Molecules, 14(2), 884-892. https://doi.org/10.3390/molecules14020884
Ranu, B. C., & Banerjee, B. (Eds.). (2020). Organoselenium Chemistry. Walter de Gruyter GmbH & Co KG.
Sonawane, A. D., & Koketsu, M. (2022). 1, 3-Selenazoles. Comprehensive Heterocyclic Chemistry IV. https://doi.org/10.1016/B978-0-12-818655-8.00089-5
Tanini, D., & Capperucci, A. (2020). Recent advances in the synthesis of five-and six-membered selena-heterocycles. Target in heterocyclic systems: chemistry and properties, 2020, 289-319.
Tao, S., Xu, L., Yang, K., Zhang, J., & Du, Y. (2022). Construction of the 2-amino-1, 3-selenazole skeleton via PhICl2/KSeCN-mediated selenocyanation/cyclization. Organic Letters, 24(23), 4187-4191. https://doi.org/10.1021/acs.orglett.2c01468
Wirth, T. (2003). Introduction and general aspects. Hypervalent Iodine Chemistry: Modern Developments in Organic Synthesis, 1-4. https://doi.org/10.1007/3-540-46114-0_1
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Biomedicine and Chemical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.



















