Biochemical Study of Some New Cephems and Selenacephems Based on 6H-1,3-Thiazines and 6H-1,3-selenazines
DOI:
https://doi.org/10.48112/bcs.v1i2.161Abstract
 Abstract Views: 447
Abstract Views: 447
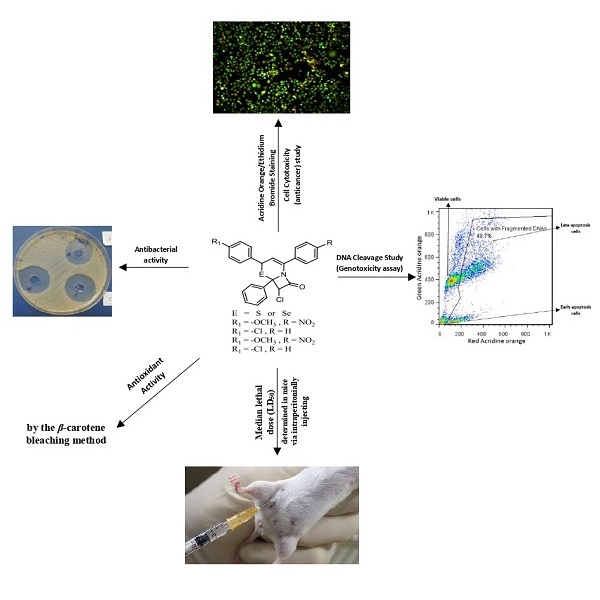
Several new and know 6-(4-substituted phenyl)-4-(4-substituted phenyl)-2-phenyl-6H-1,3-thiazine (or selenazine) (Z4B7, Z4D5, Z4B7' and Z4D5') were prepared by the 1,4-Michael addition reaction of chalcone derivatives with thiobenzamide or phenylselenocarboxamide in basic medium (where the chalcones was formed by Claisen-Schimidt condensation of aromatic aldehydes with 4-substituted acetophenone in presence of sodium hydroxide). These 6H-1,3-thia- or selenazine were used to a new series of cephem and selenacephem compounds (i.e. 7-chloro-4-(4-substituted phenyl)-2-(4-substituted phenyl)-6-phenyl-5-thia (or 5-selena)-1-azabicyclo[4.2.0]oct-2-en-8-one; AZ4B7, AZ4D5, AZ4B7' and AZ4D5'). All new compound derivatives were characterized by IR, 1H NMR, 13C NMR, mass spectroscopic techniques and elemental analysis. The toxicity of new compounds was assayed via the determination of their LD50 value by using Dixon's up and down method. The antibacterial activity of cephem and selenacephem compounds were tested in vitro against Staphylococcus aureus, Bacillus, Escherichia coli and Pseudomonas aeruginosa. Furthermore, the antioxidant, anticancer and DNA cleavage efficiency of compounds were evaluated.
Keywords:
1,3-Selenazine , 1,3-Thiazine , Biological activities , Cephem, SelenacephemReferences
Ahmeda, A., Hossain, M. A., & Ismail, Z. (2009). Antioxidant properties of the isolated flavonoids from the medicinal plant. Phyllanthus niruri. As. J. Food Ag-Ind., 2(03), 373-381.
Al-Atbi, H. S., Al-Assadi, I. J., Al-Salami, B. K., & Badr, S. Q. (2020). Study of New Azo-Azomethine Derivatives of Sulfanilamide: Synthesis, Characterization, Spectroscopic, Antimicrobial, Antioxidant and Anticancer Activity. Biochem. Cell. Arch., 20(2), 4161-4174. https://connectjournals.com/03896.2020.20.4161
Alcaide, B., Almendros, P., & Aragoncillo, C. (2007). β-Lactams: versatile building blocks for the stereoselective synthesis of non-β-lactam products. Chemical reviews, 107(11), 4437-4492. https://doi.org/10.1021/cr0307300
Al-Fartosy, A. J. (2011). Antioxidant properties of methanolic extract from Inula graveolens L. Turkish Journal of Agriculture and Forestry, 35(6), 591-596.
Al-Fregi, A. A., Al-Salami, B. K., Al-Khazragie, Z. K., & Al-Rubaie, A. Z. (2019). Synthesis, characterization and antibacterial studies of some new tellurated azo compounds. Phosphorus, Sulfur, and Silicon and the Related Elements, 194(1-2), 33-38. https://doi.org/10.1080/10426507.2018.1470179
Al-Rubaie, A. Z., Al-Masoudi, W. A., Hameed, A. J., Yousif, L. Z., & Graia, M. (2008). Synthesis, reaction and antiviral activity of 2, 4-diaryl-1, 3-selenazoles. Journal of the Korean Chemical Society, 52(1), 36-46. https://doi.org/10.5012/jkcs.2008.52.1.036
Al-Rubaie, A. Z., Yousif, L. Z., & Al-Hamad, A. J. (2002). Palladium-catalyzed formation of 3, 5-diaryl-1, 2, 4-selenadiazoles from arylselenocarboxamide. Journal of Organometallic Chemistry, 656(1-2), 274-280. https://doi.org/10.1016/S0022-328X(02)01631-5
AL-Salami, B. K., AL-Khazragie, Z. K., & Al-Fregi, A. A. (2018). Synthesis, Characterization, Antimicrobial Activity and Antioxidant of Azo Schiff Bases Containing Sulfanilamide. Journal of Global Pharma Technolog, 10(03), 952-962.
Al-Shammari, A. M., Al-Esmaeel, W. N., Al-Ali, A. A., Hassan, A. A., & Ahmed, A. A. (2019). Enhancement of Oncolytic Activity of Newcastle Disease virus Through Combination with Retinoic Acid Against Digestive System Malignancies. Molecular Therapy, 27(4S1), 126-127.
Al-Smadi, M. L., Mansour, R., Mahasneh, A., Khabour, O. F., Masadeh, M. M., & Alzoubi, K. H. (2019). Synthesis, characterization, antimicrobial activity, and genotoxicity assessment of two heterocyclic compounds containing 1, 2, 3-selena-or 1, 2, 3-thiadiazole rings. Molecules, 24(22), 4082. https://doi.org/10.3390/molecules24224082
Bairam, R., & Srinivasa, M. M. (2019). Synthesis Characterization and Biological Evaluation of Some Novel Substitute D-1, 3-Thaizine Congeners. Journal of Innovation in Pharmaceutical Sciences, 3(1), 33-39.
Baldo, B. A., Zhao, Z., & Pham, N. H. (2008). Antibiotic allergy: immunochemical and clinical considerations. Current Allergy and Asthma Reports, 8(1), 49-55. https://doi.org/10.1007/s11882-008-0010-1
Bartzatt, R. (2016). Acridine Orange Staining For Identifying Viral Infection of Cells In-Vitro and Cellular DNA. ChemXpress, 9(5), 102.
Bhalla, A., Bari, S. S., Berry, S., Bhalla, J., Vats, S., Mandal, S., & Khullarb, S. (2015). Facile synthesis of novel monocyclic trans-and cis-3-oxy/thio/seleno-4-pyrazolyl-β-lactams. ARKIVOC, 7, 10-27.
Bhattacharjee, M. K. (2016). Chemistry of antibiotics and related drugs (Vol. 8). Cham: Springer.
Bhupendra, M., & Smita, J. (2010). Synthesis and characterization of some quinoline based azetidinones and thiazolidinones as antimicrobial agents. Archives of Applied Science Research, 2(6), 332-343.
Broccolo, F., Cainelli, G., Caltabiano, G., Cocuzza, C. E., Fortuna, C. G., Galletti, P., ... & Quintavalla, A. (2006). Design, synthesis, and biological evaluation of 4-alkyliden-beta lactams: new products with promising antibiotic activity against resistant bacteria. Journal of medicinal chemistry, 49(9), 2804-2811. https://doi.org/10.1021/jm0580510
Dalhoff, A., & Thomson, C. J. (2003). The art of fusion: from penams and cephems to penems. Chemotherapy, 49(3), 105-120. https://doi.org/10.1159/000070616
Elkanzi, N. A. A. (2013). Short review on synthesis of thiazolidinone and β-lactam. World, 1(2), 24-51.
El-Sherif, A. A., & Eldebss, T. M. (2011). Synthesis, spectral characterization, solution equilibria, in vitro antibacterial and cytotoxic activities of Cu (II), Ni (II), Mn (II), Co (II) and Zn (II) complexes with Schiff base derived from 5-bromosalicylaldehyde and 2-aminomethylthiophene. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 79(5), 1803-1814. https://doi.org/10.1016/j.saa.2011.05.062
Freshney, R. I. (2010). Culture of animal cells a manual of basic technique and specialized applications, 6th Ed., Wiley-Blackwell, 732.
Friot, C., Reliquet, A., & Meslin, J. C. (1997). Synthese De Cephemes Par Reaction De Nouvelles 6H-1, 3-Thiazines Avec Les Cetenes. Phosphorus, Sulfur, and Silicon and the Related Elements, 131(1), 147-160. https://doi.org/10.1080/10426509708031603
Fuentes, A., Marinas, J. M., & Sinisterra, J. V. (1987). Catalyzed synthesis of chalcones under interfacial solid-liquid conditions with ultrasound. Tetrahedron Letters, 28(39), 4541-4544. https://doi.org/10.1016/S0040-4039(00)96558-4
Hejchman, E., Kruszewska, H., Maciejewska, D., Sowirka-Taciak, B., Tomczyk, M., Sztokfisz-Ignasiak, A., ... & Młynarczuk-Biały, I. (2019). Design, synthesis, and biological activity of Schiff bases bearing salicyl and 7-hydroxycoumarinyl moieties. Monatshefte für Chemie-Chemical Monthly, 150(2), 255-266. https://doi.org/10.1007/s00706-018-2325-5
Hossain, M. M., Aziz, M. D., Ahmed, R., Hossain, M., Mahmud, A., Ahmed, T., & Mazumder, E. H. (2010). In Vitro Free Radical Scavenging Activity of Some βLactams And Phenolics. International Journal of Pharmacy and Pharmaceutical Sciences, 2(2).
Katiyar, R. S., Singhvi, N. R., Kushwaha, R. V., Ramji, Lal., & Suryanarayana, N. V. (2009). A mycorrhizal association in arjuna and jamun trees in forest of Bhandara region, Maharashtra, India. International Journal of Agricultural Sciences, 4, 229-232.
Kazi, A., Hill, R., Long, T. E., Kuhn, D. J., Turos, E., & Dou, Q. P. (2004). Novel N-thiolated β-lactam antibiotics selectively induce apoptosis in human tumor and transformed, but not normal or nontransformed, cells. Biochemical pharmacology, 67(2), 365-374. https://doi.org/10.1016/j.bcp.2003.09.017
Kuhn, D., Coates, C., Daniel, K., Chen, D., Bhuiyan, M., Kazi, A., ... & Dou, Q. P. (2004). Beta-lactams and their potential use as novel anticancer chemotherapeutics drugs. Front Biosci, 9, 2605-2617.
Liu, K., Liu, P. C., Liu, R., & Wu, X. (2015). Dual AO/EB staining to detect apoptosis in osteosarcoma cells compared with flow cytometry. Medical science monitor basic research, 21, 15. https://dx.doi.org/10.12659%2FMSMBR.893327
Miladi, S., & Damak, M. (2008). "In Vitro Antioxidant A ctivities of Aloe vera Leaf Skin Extracts''. Journal of Society of Chemstry, Tunisie, 10, 101-109.
Mohana, K. N., & Kumar, C. B. P. (2013). Synthesis and antioxidant activity of 2-amino-5-methylthiazol derivatives containing 1, 3, 4-oxadiazole-2-thiol moiety. International Scholarly Research Notices, 2013.
Mukhtar, H. (2012). Chemoprevention: making it a success story for controlling human cancer. Cancer letters, 326(2), 123-127. https://doi.org/10.1016/j.canlet.2012.05.016
Muratori, M., Tamburrino, L., Marchiani, S., Cambi, M., Olivito, B., Azzari, C., ... & Baldi, E. (2015). Investigation on the origin of sperm DNA fragmentation: role of apoptosis, immaturity and oxidative stress. Molecular medicine, 21(1), 109-122. https://doi.org/10.2119/molmed.2014.00158
Ninomiya, M., Garud, D. R., & Koketsu, M. (2011). Biologically significant selenium-containing heterocycles. Coordination Chemistry Reviews, 255(23-24), 2968-2990. https://doi.org/10.1016/j.ccr.2011.07.009
Nishio, T., & Ori, M. (2001). Thionation of ω‐Acylamino Ketones with Lawesson's Reagent: Convenient Synthesis of 1, 3‐Thiazoles and 4H‐1, 3‐Thiazines. Helvetica Chimica Acta, 84(8), 2347-2354. https://doi.org/10.1002/1522-2675(20010815)84:8<2347::AID-HLCA2347>3.0.CO;2-N
Norrihan, S., Md Abu, A., Md Abdus, S., Fasihuddin B, A., & Mohd Razip, A. (2012). Synthesis, spectral characterization and biological activities of Organotin (IV) complexes with ortho-vanillin-2-hydrazinopyridine (VHP). Open Journal of Inorganic Chemistry, 2012. http://www.scirp.org/journal/PaperInformation.aspx?PaperID=18652
Patil, P. S., Teh, J. J., Fun, H. K., Razak, I. A., & Dharmaprakash, S. M. (2006). 3-(4-Methoxyphenyl)-1-(4-nitrophenyl) prop-2-en-1-one. Acta Crystallographica Section E: Structure Reports Online, 62(3), o896-o898. https://doi.org/10.1107/S1600536806003564
Piewngam, P., & Otto, M. (2020). Probiotics to prevent Staphylococcus aureus disease?. Gut Microbes, 11(1), 94-101. https://doi.org/10.1080/19490976.2019.1591137
Rezaei, M., Komijani, M., & Javadirad, S. M. (2012). Bacteriostatic Agents. In A Search for Antibacterial Agents. IntechOpen.
Rispin, A., Farrar, D., Margosches, E., Gupta, K., Stitzel, K., Carr, G., ... & McCall, D. (2002). Alternative methods for the median lethal dose (LD50) test: the up-and-down procedure for acute oral toxicity. ILAR journal, 43(4), 233-243. https://doi.org/10.1093/ilar.43.4.233
Schmidt, R. R., & Dimmler, M. (1975). Heterocyclische 8π‐Systeme, 5. 1, 3‐Thiazin‐Anionen. Chemische Berichte, 108(1), 6-16. https://doi.org/10.1002/cber.19751080103
Sharp, D. J., Rogers, G. C., & Scholey, J. M. (2000). Microtubule motors in mitosis. Nature, 407(6800), 41-47. https://doi.org/10.1038/35024000
Smania Jr, A., Monache, F. D., Smania, E. D. F. A., & Cuneo, R. S. (1999). Antibacterial activity of steroidal compounds isolated from Ganoderma applanatum (Pers.) Pat.(Aphyllophoromycetideae) fruit body. International Journal of medicinal mushrooms, 1(4). https://doi.org/10.1615/INTJMEDMUSHR.V1.I4.40
Smith, D. M., Kazi, A., Smith, L., Long, T. E., Heldreth, B., Turos, E., & Dou, Q. P. (2002). A novel β-lactam antibiotic activates tumor cell apoptotic program by inducing DNA damage. Molecular pharmacology, 61(6), 1348-1358. https://doi.org/10.1124/mol.61.6.1348
Sommen, G. L., Linden, A., & Heimgartner, H. (2005). Selenium-Containing Heterocycles From Isoselenocyanates: Synthesis of 1, 3-Selenazolidine and Perhydro-1, 3-selenazine Derivatives. European Journal of Organic Chemistry, (14), 3128-3137. https://doi.org/10.1002/ejoc.200500090
Usova, E. B., Krapivin, G. D., Zavodnik, V. E., & Kul'nevich, V. G. (1994). Synthesis and properties of 5-furyl (aryl)-Δ2-1, 2, 4-triazolines and-Δ2-1, 3, 4-thiadiazolines. Molecular and crystal structure of 2-acetylamino-5-phenyl-Δ2-1, 3, 4-thiadiazoline. Chemistry of Heterocyclic Compounds, 30(10), 1158-1164. https://doi.org/10.1007/BF01184877
van Zandwijk, N. (2005). Chemoprevention in lung carcinogenesis–an overview. European journal of cancer, 41(13), 1990-2002. https://doi.org/10.1016/j.ejca.2005.05.011
Verdino, A., Vigliotta, G., Giordano, D., Caputo, I., Soriente, A., De Rosa, M., & Marabotti, A. (2017). Synthesis and biological evaluation of the progenitor of a new class of cephalosporin analogues, with a particular focus on structure-based computational analysis. PloS one, 12(7), e0181563. https://doi.org/10.1371/journal.pone.0181563
Xia, Y., Liu, X., Zhang, L., Zhang, J., Li, C., Zhang, N., ... & Li, Y. (2019). A new Schiff base coordinated copper (II) compound induces apoptosis and inhibits tumor growth in gastric cancer. Cancer Cell International, 19(1), 1-11. https://doi.org/10.1186/s12935-019-0801-6
Zini, A., & Agarwal, A. (Eds.). (2011). Sperm chromatin: biological and clinical applications in male infertility and assisted reproduction (p. 512). New York: Springer.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Biomedicine and Chemical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.



















