Metabolic Engineering of Microbial Cell Factories for Sustainable Biomanufacturing
DOI:
https://doi.org/10.5281/zenodo.15287978Abstract
 Abstract Views: 425
Abstract Views: 425
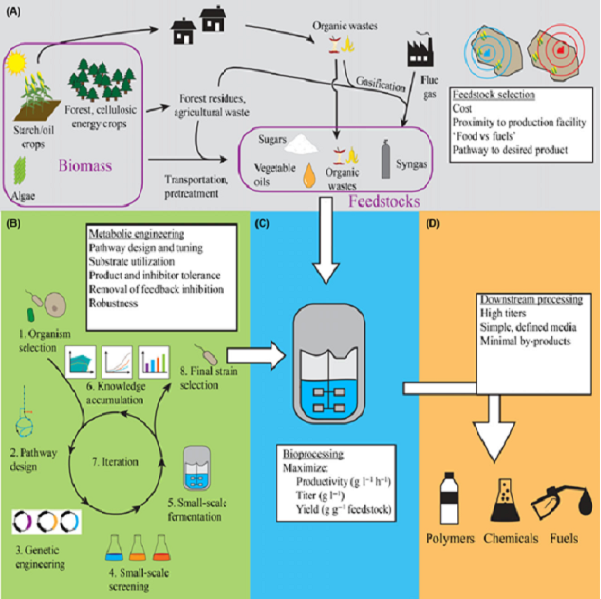
Metabolic engineering is essential for the development of microbial cell factories to produce biomolecules from low-value renewable substrates. This role helps advance the development of ecologically responsible and commercially robust chemical industries, including biofuels and high-value compounds like medicines. The ability of microbial cell factories to generate a wide variety of substances sustainably, therefore satisfying modern commodity needs, has piqued the scientific community's attention. The goal of metabolic engineering is to convert different microorganisms into efficient cell factories to produce desired products, and it has been used for decades to develop novel metabolic pathways and alter pre-existing ones with the help of system biology, synthetic biology, and evolutionary engineering.
Keywords:
Biofuels, Cell factories, Metabolic engineering , Microbial cell, Microorganisms, Synthetic biologyReferences
Koffas, M., Roberge, C., Lee, K., & Stephanopoulos, G. (1999). Metabolic Engineering. Annual Review of Biomedical Engineering, 1(1), 535–557. https://doi.org/10.1146/annurev.bioeng.1.1.535
Lee, G., Lee, S. M., & Kim, H. U. (2023). A contribution of metabolic engineering to addressing medical problems: Metabolic flux analysis. Metabolic Engineering, 77, 283–293. https://doi.org/10.1016/j.ymben.2023.04.008
Rahmat, E., & Kang, Y. (2020). Yeast metabolic engineering for the production of pharmaceutically important secondary metabolites. Applied Microbiology and Biotechnology, 104(11), 4659–4674. https://doi.org/10.1007/s00253-020-10587-y
Munro, L. J., & Kell, D. B. (2021). Intelligent host engineering for metabolic flux optimisation in biotechnology. The Biochemical Journal, 478(20), 3685–3721. https://doi.org/10.1042/BCJ20210535
Onda, M., Vincent, J. J., Lee, B., & Pastan, I. (2003). Mutants of Immunotoxin Anti-Tac(dsFv)-PE38 with Variable Number of Lysine Residues as Candidates for Site-Specific Chemical Modification. 1. Properties of Mutant Molecules. Bioconjugate Chemistry, 14(2), 480–487. https://doi.org/10.1021/bc020069r
Withers, S. T., & Keasling, J. D. (2007). Biosynthesis and engineering of isoprenoid small molecules. Applied Microbiology and Biotechnology, 73(5), 980–990. https://doi.org/10.1007/s00253-006-0593-1
Watstein, D. M., McNerney, M. P., & Styczynski, M. P. (2015). Precise metabolic engineering of carotenoid biosynthesis in Escherichia coli towards a low-cost biosensor. Metabolic Engineering, 31, 171–180. https://doi.org/10.1016/j.ymben.2015.06.007
Benner, S. A., & Sismour, A. M. (2005). Synthetic biology. Nature Reviews Genetics, 6(7), 533–543. https://doi.org/10.1038/nrg1637
Andrianantoandro, E., Basu, S., Karig, D. K., & Weiss, R. (2006). Synthetic biology: New engineering rules for an emerging discipline. Molecular Systems Biology, 2(1), 2006.0028. https://doi.org/10.1038/msb4100073
Shih, P. M., Liang, Y., & Loque, D. (2016). Biotechnology and synthetic biology approaches for metabolic engineering of bioenergy crops. The Plant Journal, 87(1), 103-117. https://doi.org/10.1111/tpj.13176
Lee, J. W., Na, D., Park, J. M., Lee, J., Choi, S., & Lee, S. Y. (2012). Systems metabolic engineering of microorganisms for natural and non-natural chemicals. Nature Chemical Biology, 8(6), 536–546. https://doi.org/10.1038/nchembio.970
Choi, K. R., Jang, W. D., Yang, D., Cho, J. S., Park, D., & Lee, S. Y. (2019). Systems metabolic engineering strategies: Integrating systems and synthetic biology with metabolic engineering. Trends in Biotechnology, 37(8), 817–837. https://doi.org/10.1016/j.tibtech.2019.01.003
Acevedo-Rocha, C. G., Gronenberg, L. S., Mack, M., Commichau, F. M., & Genee, H. J. (2019). Microbial cell factories for the sustainable manufacturing of B vitamins. Food Biotechnology • Plant Biotechnology, 56, 18–29. https://doi.org/10.1016/j.copbio.2018.07.006
Yang, Z., Liang, G., & Xu, B. (2008). Enzymatic hydrogelation of small molecules. Accounts of Chemical Research, 41(2), 315–326. https://doi.org/10.1021/ar7001914
Rabara, R. C., Tripathi, P., & Rushton, P. J. (2014). The potential of transcription factor-based genetic engineering in improving crop tolerance to drought. Omics: A Journal of Integrative Biology, 18(10), 601–614. https://doi.org/10.1089/omi.2013.0177
Lee, S. Y., & Kim, H. U. (2015). Systems strategies for developing industrial microbial strains. Nature Biotechnology, 33(10), 1061–1072. https://doi.org/10.1038/nbt.3365
Nevoigt, E. (2008). Progress in metabolic engineering of Saccharomyces cerevisiae. Microbiology and Molecular Biology Reviews, 72(3), 379–412. https://doi.org/10.1128/mmbr.00025-07
Salazar, J., & Meil, J. (2009). Prospects for carbon-neutral housing: The influence of greater wood use on the carbon footprint of a single-family residence. Journal of Cleaner Production, 17(17), 1563–1571. https://doi.org/10.1016/j.jclepro.2009.06.006
Lowry, G. V., Gregory, K. B., Apte, S. C., & Lead, J. R. (2012). Transformations of Nanomaterials in the Environment. Environmental Science & Technology, 46(13), 6893–6899. https://doi.org/10.1021/es300839e
Ko, Y.-S., Kim, J. W., Lee, J. A., Han, T., Kim, G. B., Park, J. E., & Lee, S. Y. (2020). Tools and strategies of systems metabolic engineering for the development of microbial cell factories for chemical production. Chemical Society Reviews, 49(14), 4615–4636. https://doi.org/10.1039/d0cs00155d
Yoon, J. M., Zhao, L., & Shanks, J. V. (2013). Metabolic engineering with plants for a sustainable biobased economy. Annual Review of Chemical and Biomolecular Engineering, 4, 211–237. https://doi.org/10.1146/annurev-chembioeng-061312-103320
Choi, S. Y., Cho, I. J., Lee, Y., Kim, Y.-J., Kim, K.-J., & Lee, S. Y. (2020). Microbial Polyhydroxyalkanoates and Nonnatural Polyesters. Advanced Materials, 32(35), 1907138. https://doi.org/10.1002/adma.201907138
Schuster, S., Fell, D. A., & Dandekar, T. (2000). A general definition of metabolic pathways useful for systematic organization and analysis of complex metabolic networks. Nature Biotechnology, 18(3), 326–332. https://doi.org/10.1038/73786
Bilal, M., Iqbal, H. M., Guo, S., Hu, H., Wang, W., & Zhang, X. (2018). State-of-the-art protein engineering approaches using biological macromolecules: A review from immobilization to implementation view point. International Journal of Biological Macromolecules, 108, 893–901. https://doi.org/10.1016/j.ijbiomac.2017.10.182
Beger, R. D., Dunn, W., Schmidt, M. A., Gross, S. S., Kirwan, J. A., Cascante, M., Brennan, L., Wishart, D. S., Oresic, M., & Hankemeier, T. (2016). Metabolomics enables precision medicine:“a white paper, community perspective”. Metabolomics, 12, 1–15. https://doi.org/10.1007/s11306-016-1094-6
Cravens, A., Payne, J., & Smolke, C. D. (2019). Synthetic biology strategies for microbial biosynthesis of plant natural products. Nature Communications, 10(1), 2142. https://doi.org/10.1038/s41467-019-09848-w
Rollié, S., Mangold, M., & Sundmacher, K. (2012). Designing biological systems: Systems engineering meets synthetic biology. Chemical Engineering Science, 69(1), 1–29. https://doi.org/10.1016/j.ces.2011.10.068
Walker, R. S. K., & Pretorius, I. S. (2018). Applications of Yeast Synthetic Biology Geared towards the Production of Biopharmaceuticals. Genes, 9(7). https://doi.org/10.3390/genes9070340
Hibbert, E. G., & Dalby, P. A. (2005). Directed evolution strategies for improved enzymatic performance. Microbial Cell Factories, 4(1), 29. https://doi.org/10.1186/1475-2859-4-29
Jendoubi, T. (2021). Approaches to Integrating Metabolomics and Multi-Omics Data: A Primer. Metabolites, 11(3). https://doi.org/10.3390/metabo11030184
Cho, J. S., Kim, G. B., Eun, H., Moon, C. W., & Lee, S. Y. (2022). Designing Microbial Cell Factories for the Production of Chemicals. JACS Au, 2(8), 1781–1799. https://doi.org/10.1021/jacsau.2c00344
Zhao, L., Zhu, Y., Jia, H., Han, Y., Zheng, X., Wang, M., & Feng, W. (2022). From Plant to Yeast-Advances in Biosynthesis of Artemisinin. Molecules (Basel, Switzerland), 27(20). https://doi.org/10.3390/molecules27206888
Pu, W., Chen, J., Zhou, Y., Qiu, H., Shi, T., Zhou, W., Guo, X., Cai, N., Tan, Z., Liu, J., Feng, J., Wang, Y., Zheng, P., & Sun, J. (2023). Systems metabolic engineering of Escherichia coli for hyper-production of 5‑aminolevulinic acid. Biotechnology for Biofuels and Bioproducts, 16(1), 31. https://doi.org/10.1186/s13068-023-02280-9
Hellmuth, K. (2006). Industrial scale production of chymosin with Aspergillus niger. Microbial Cell Factories, 5(1), S31. https://doi.org/10.1186/1475-2859-5-S1-S31
Sehgal, R., & Gupta, R. (2020). Polyhydroxyalkanoate and its efficient production: An eco-friendly approach towards development. 3 Biotech, 10(12), 549. https://doi.org/10.1007/s13205-020-02550-5
Zhu, X., Liu, X., Liu, T., Wang, Y., Ahmed, N., Li, Z., & Jiang, H. (2021). Synthetic biology of plant natural products: From pathway elucidation to engineered biosynthesis in plant cells. Plant Communications, 2(5), 100229. https://doi.org/10.1016/j.xplc.2021.100229
Hommel, M. (2008). The future of artemisinins: Natural, synthetic or recombinant? Journal of Biology, 7(10), 38. https://doi.org/10.1186/jbiol101
Paddon, C. J., Westfall, P. J., Pitera, D. J., Benjamin, K., Fisher, K., McPhee, D., Leavell, M. D., Tai, A., Main, A., Eng, D., Polichuk, D. R., Teoh, K. H., Reed, D. W., Treynor, T., Lenihan, J., Jiang, H., Fleck, M., Bajad, S., Dang, G., … Newman, J. D. (2013). High-level semi-synthetic production of the potent antimalarial artemisinin. Nature, 496(7446), 528–532. https://doi.org/10.1038/nature12051
Luerce, T. D., Azevedo, M. S. P., LeBlanc, J. G., Azevedo, V., Miyoshi, A., & Pontes, D. S. (2014). Recombinant Lactococcus lactis fails to secrete bovine chymosine. Bioengineered, 5(6), 363–370. https://doi.org/10.4161/bioe.36327
Silano, V., Barat Baviera, J. M., Bolognesi, C., Cocconcelli, P. S., Crebelli, R., Gott, D. M., Grob, K., Lambré, C., Lampi, E., Mengelers, M., Mortensen, A., Rivière, G., Steffensen, I.-L., Tlustos, C., Van Loveren, H., Vernis, L., Zorn, H., Aguilera, J., Andryszkiewicz, M., … Chesson, A. (2022). Safety evaluation of the food enzyme chymosin from the genetically modified Aspergillus niger strain DSM 29544. EFSA Journal. European Food Safety Authority, 20(8), e07464. https://doi.org/10.2903/j.efsa.2022.7464
Pickens, L. B., Tang, Y., & Chooi, Y.-H. (2011). Metabolic engineering for the production of natural products. Annual Review of Chemical and Biomolecular Engineering, 2, 211–236. https://doi.org/10.1146/annurev-chembioeng-061010-114209
Wei, Z.-Y., Zhang, Y.-Y., Wang, Y.-P., Fan, M.-X., Zhong, X.-F., Xu, N., Lin, F., & Xing, S.-C. (2016). Production of Bioactive Recombinant Bovine Chymosin in Tobacco Plants. International Journal of Molecular Sciences, 17(5). https://doi.org/10.3390/ijms17050624
Prache, S., Adamiec, C., Astruc, T., Baéza-Campone, E., Bouillot, P. E., Clinquart, A., Feidt, C., Fourat, E., Gautron, J., Girard, A., Guillier, L., Kesse-Guyot, E., Lebret, B., Lefèvre, F., Le Perchec, S., Martin, B., Mirade, P. S., Pierre, F., Raulet, M., … Santé-Lhoutellier, V. (2022). Review: Quality of animal-source foods. Quality of Animal-Source Foods, 16, 100376. https://doi.org/10.1016/j.animal.2021.100376
Daboussi, F., & Lindley, N. D. (2023). Challenges to Ensure a Better Translation of Metabolic Engineering for Industrial Applications. Methods in Molecular Biology (Clifton, N.J.), 2553, 1–20. https://doi.org/10.1007/978-1-0716-2617-7_1
Tomé, D., & Bos, C. (2007). Lysine Requirement through the Human Life Cycle. The Journal of Nutrition, 137(6), 1642S-1645S. https://doi.org/10.1093/jn/137.6.1642S
Liao, S. F., Wang, T., & Regmi, N. (2015). Lysine nutrition in swine and the related monogastric animals: Muscle protein biosynthesis and beyond. SpringerPlus, 4, 147. https://doi.org/10.1186/s40064-015-0927-5
Jakobsen, O. M., Brautaset, T., Degnes, K. F., Heggeset, T. M. B., Balzer, S., Flickinger, M. C., Valla, S., & Ellingsen, T. E. (2009). Overexpression of wild-type aspartokinase increases L-lysine production in the thermotolerant methylotrophic bacterium Bacillus methanolicus. Applied and Environmental Microbiology, 75(3), 652–661. https://doi.org/10.1128/AEM.01176-08
Hallen, A., Jamie, J. F., & Cooper, A. J. L. (2013). Lysine metabolism in mammalian brain: An update on the importance of recent discoveries. Amino Acids, 45(6), 1249–1272. https://doi.org/10.1007/s00726-013-1590-1
Korosh, T. C., Markley, A. L., Clark, R. L., McGinley, L. L., McMahon, K. D., & Pfleger, B. F. (2017). Engineering photosynthetic production of L-lysine. Metabolic Engineering, 44, 273–283. https://doi.org/10.1016/j.ymben.2017.10.010
Calero, P., & Nikel, P. I. (2019). Chasing bacterial chassis for metabolic engineering: A perspective review from classical to non-traditional microorganisms. Microbial Biotechnology, 12(1), 98–124. https://doi.org/10.1111/1751-7915.13292
Rasor, B. J., Karim, A. S., Alper, H. S., & Jewett, M. C. (2023). Cell Extracts from Bacteria and Yeast Retain Metabolic Activity after Extended Storage and Repeated Thawing. ACS Synthetic Biology, 12(3), 904–908. https://doi.org/10.1021/acssynbio.2c00685
Vitorino, L. C., & Bessa, L. A. (2017). Technological Microbiology: Development and Applications. Frontiers in Microbiology, 8, 827. https://doi.org/10.3389/fmicb.2017.00827
Ullah, N., Shahzad, K., & Wang, M. (2021). The Role of Metabolic Engineering Technologies for the Production of Fatty Acids in Yeast. Biology, 10(7). https://doi.org/10.3390/biology10070632
Yuan, S.-F., & Alper, H. S. (2019). Metabolic engineering of microbial cell factories for production of nutraceuticals. Microbial Cell Factories, 18(1), 46. https://doi.org/10.1186/s12934-019-1096-y
Bajić, B., Vučurović, D., Vasić, Đ., Jevtić-Mučibabić, R., & Dodić, S. (2022). Biotechnological Production of Sustainable Microbial Proteins from Agro-Industrial Residues and By-Products. Foods (Basel, Switzerland), 12(1). https://doi.org/10.3390/foods12010107
Zhang, M. M., Wang, Y., Ang, E. L., & Zhao, H. (2016). Engineering microbial hosts for production of bacterial natural products. Natural Product Reports, 33(8), 963–987. https://doi.org/10.1039/c6np00017g
Wang, F., Harindintwali, J. D., Yuan, Z., Wang, M., Wang, F., Li, S., Yin, Z., Huang, L., Fu, Y., Li, L., Chang, S. X., Zhang, L., Rinklebe, J., Yuan, Z., Zhu, Q., Xiang, L., Tsang, D. C. W., Xu, L., Jiang, X., … Chen, J. M. (2021). Technologies and perspectives for achieving carbon neutrality. The Innovation, 2(4), 100180. https://doi.org/10.1016/j.xinn.2021.100180
Zheng, B., Yu, S., Chen, Z., & Huo, Y.-X. (2022). A consolidated review of commercial-scale high-value products from lignocellulosic biomass. Frontiers in Microbiology, 13. https://www.frontiersin.org/articles/10.3389/fmicb.2022.933882
Dai, X., & Shen, L. (2022). Advances and trends in omics technology development. Frontiers in Medicine, 9, 911861. https://doi.org/10.3389/fmed.2022.911861
Van Dien, S. (2013). From the first drop to the first truckload: Commercialization of microbial processes for renewable chemicals. Current Opinion in Biotechnology, 24(6), 1061–1068. https://doi.org/10.1016/j.copbio.2013.03.002
Müller, D., Klein, L., Lemke, J., Schulze, M., Kruse, T., Saballus, M., Matuszczyk, J., Kampmann, M., & Zijlstra, G. (2022). Process intensification in the biopharma industry: Improving efficiency of protein manufacturing processes from development to production scale using synergistic approaches. Chemical Engineering and Processing - Process Intensification, 171, 108727. https://doi.org/10.1016/j.cep.2021.108727
Croughan, M. S., Konstantinov, K. B., & Cooney, C. (2015). The future of industrial bioprocessing: Batch or continuous? Biotechnology and Bioengineering, 112(4), 648–651. https://doi.org/10.1002/bit.25529
Poontawee, R., & Limtong, S. (2020). Feeding Strategies of Two-Stage Fed-Batch Cultivation Processes for Microbial Lipid Production from Sugarcane Top Hydrolysate and Crude Glycerol by the Oleaginous Red Yeast Rhodosporidiobolus fluvialis. Microorganisms, 8(2). https://doi.org/10.3390/microorganisms8020151
Kim, W.-T., & Ryu, C. J. (2017). Cancer stem cell surface markers on normal stem cells. BMB Reports, 50(6), 285. https://doi.org/10.5483/bmbrep.2017.50.6.039
HSU, S. Y. S. (2020). Rational Engineering of Expression Level of Multi-Gene Systems encoding Natural Product Biosynthesis in Streptomyces (Doctoral dissertation, University of Minnesota).
Baumann, P., & Hubbuch, J. (2017). Downstream process development strategies for effective bioprocesses: Trends, progress, and combinatorial approaches. Engineering in Life Sciences, 17(11), 1142–1158. https://doi.org/10.1002/elsc.201600033
Ankit, Saha, L., Kumar, V., Tiwari, J., Sweta, Rawat, S., Singh, J., & Bauddh, K. (2021). Electronic waste and their leachates impact on human health and environment: Global ecological threat and management. Environmental Technology & Innovation, 24, 102049. https://doi.org/10.1016/j.eti.2021.102049
Yang, M., Yun, J., Zhang, H., Magocha, T. A., Zabed, H., Xue, Y., ... & Qi, X. (2018). Genetically engineered strains: application and advances for 1, 3-propanediol production from glycerol. Food technology and biotechnology, 56(1), 3–15. https://doi.org/10.17113/ftb.56.01.18.5444
Gorden, J., von Helden, E., Wierckx, N., Blank, L., Zeiner, T., & Brandenbusch, C. (2017). Integrated process development of a reactive extraction concept for itaconic acid and application to a real fermentation broth. Engineering in Life Sciences, 17. https://doi.org/10.1002/elsc.201600253
Siew, Y. Y., & Zhang, W. (2021). Downstream processing of recombinant human insulin and its analogues production from E. coli inclusion bodies. Bioresources and Bioprocessing, 8(1), 65. https://doi.org/10.1186/s40643-021-00419-w
Casado López, S., Peng, M., Issak, T. Y., Daly, P., de Vries, R. P., & Mäkelä, M. R. (2018). Induction of Genes Encoding Plant Cell Wall-Degrading Carbohydrate-Active Enzymes by Lignocellulose-Derived Monosaccharides and Cellobiose in the White-Rot Fungus Dichomitus squalens. Applied and Environmental Microbiology, 84(11). https://doi.org/10.1128/AEM.00403-18
Geijer, C., Ledesma-Amaro, R., & Tomás-Pejó, E. (2022). Unraveling the potential of non-conventional yeasts in biotechnology. FEMS Yeast Research, 22(1). https://doi.org/10.1093/femsyr/foab071
Du, J., Shao, Z., & Zhao, H. (2011). Engineering microbial factories for synthesis of value-added products. Journal of Industrial Microbiology & Biotechnology, 38(8), 873–890. https://doi.org/10.1007/s10295-011-0970-3
Parapouli, M., Vasileiadis, A., Afendra, A.-S., & Hatziloukas, E. (2020). Saccharomyces cerevisiae and its industrial applications. AIMS Microbiology, 6(1), 1–31. https://doi.org/10.3934/microbiol.2020001
Lande, R. (1976). Natural Selection and Random Genetic Drift in Phenotypic Evolution. Evolution, 30(2), 314–334. JSTOR. https://doi.org/10.2307/2407703
Reygaert, W. C. (2018). An overview of the antimicrobial resistance mechanisms of bacteria. AIMS Microbiology, 4(3), 482–501. https://doi.org/10.3934/microbiol.2018.3.482
Balan, V. (2014). Current challenges in commercially producing biofuels from lignocellulosic biomass. ISRN Biotechnology, 2014, 463074. https://doi.org/10.1155/2014/463074
Wackett, L. P. (2008). Microbial-based motor fuels: Science and technology. Microbial Biotechnology, 1(3), 211–225. https://doi.org/10.1111/j.1751-7915.2007.00020.x
Liang, F.-Y., Ryvak, M., Sayeed, S., & Zhao, N. (2012). The role of natural gas as a primary fuel in the near future, including comparisons of acquisition, transmission and waste handling costs of as with competitive alternatives. Chemistry Central Journal, 6 Suppl 1(Suppl 1), S4. https://doi.org/10.1186/1752-153X-6-S1-S4
Manfroni, M., Bukkens, S. G. F., & Giampietro, M. (2022). Securing fuel demand with unconventional oils: A metabolic perspective. Energy, 261, 125256. https://doi.org/10.1016/j.energy.2022.125256
Chen, G.-Q. (2009). A microbial polyhydroxyalkanoates (PHA) based bio-and materials industry. Chemical Society Reviews, 38(8), 2434–2446. https://doi.org/10.1039/b812677c
Chen, H., Doni, S., & Masciandaro, G. Assessment of Biodegradation in Different Environmental Compartments of Blends and Composites Based on Microbial Poly (hydroxyalkanoate) s. Pisa Univ. Pisa, 1–191.
Montaño López, J., Duran, L., & Avalos, J. L. (2022). Physiological limitations and opportunities in microbial metabolic engineering. Nature Reviews Microbiology, 20(1), 35–48. https://doi.org/10.1038/s41579-021-00600-0
Fletcher, E., Chen, Y., Caspeta, L., & Feizi, A. (2022). Editorial: Genomic strategies for efficient microbial cell factories. Frontiers in Bioengineering and Biotechnology, 10. https://doi.org/10.3389/fbioe.2022.962828
Liu, T., Xu, X., Liu, Y., Li, J., Du, G., Lv, X., & Liu, L. (2022). Engineered Microbial Cell Factories for Sustainable Production of L-Lactic Acid: A Critical Review. Fermentation, 8(6). https://doi.org/10.3390/fermentation8060279
Noseda, D. G., Recúpero, M. N., Blasco, M., Ortiz, G. E., & Galvagno, M. A. (2013). Cloning, expression and optimized production in a bioreactor of bovine chymosin B in Pichia (Komagataella) pastoris under AOX1 promoter. Protein Expression and Purification, 92(2), 235–244. https://doi.org/10.1016/j.pep.2013.08.018
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Biomedicine and Chemical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.



















