Histological Changes of Liver Tissue Triggered by Antibiotic Drug Azithromycin and the Ameliorative Effects of Garlic in Male Albino Rats
DOI:
https://doi.org/10.5281/zenodo.15778025Abstract
 Abstract Views: 180
Abstract Views: 180
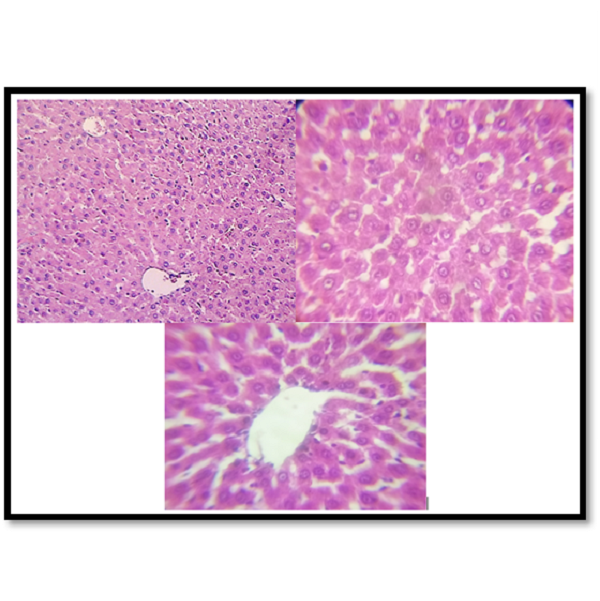
The garlic plant can be used as a spice in cooking or as a treatment for several common disorders. Garlic's four primary constituents are allicin, alliin, diallyl sulfide (DAS), and S-allyl cysteine (SAC). Anti-inflammatory, anti-cancer, antidiabetic, and antioxidant qualities are among its biological attributes. By using histological analysis of liver tissue, the work aimed to determine the harmful effects of the antibiotic Azithromycin, as well as the protective function of garlic. In the animal house of the Faculty of Science, University of Kufa, 24 mature male albino rats (Rattus norvegicus) weighing between 180 and 270 grams on average were employed in this study. The rats were between 2.5 and 3 months old. According to the study, two weeks after oral administration of 500 mg/kg of azithromycin and 240 mg/kg of garlic to animals, histological changes in liver tissue were estimated. Compared to the control group, which acquired distilled water and garlic, several negative effects on liver tissue were observed in the groups that received azithromycin and azithromycin + garlic. Furthermore, compared to the group that was given azithromycin alone, the results displayed a minor alteration in liver tissue in the group that received azithromycin plus garlic.
Keywords:
Azithromycin, Garlic, Histological changes, Liver damageReferences
Aly, S. M., Fetaih, H. A., Hassanin, A. A., Abomughaid, M. M., & Ismail, A. A. (2019). Protective effects of garlic and cinnamon oils on hepatocellular carcinoma in albino rats. Analytical Cellular Pathology, 2019(1), 9895485. https://doi.org/10.1155/2019/9895485
Bancroft, J. D., & Gamble, M. (Eds.). (2008). Theory and practice of histological techniques. Elsevier Health Sciences.
Dabdoub, B. R., & Abdulhadi, H. L. (2022). Hepatoprotective effect of luteolin against azithromycin induced hepatotoxicity. Jundishapur J. Microbiol, 15(1), 1417.
Doan, T., Hinterwirth, A., Arzika, A. M., Cotter, S. Y., Ray, K. J., O’Brien, K. S., ... & Lietman, T. M. (2018, August). Mass azithromycin distribution and community microbiome: a cluster-randomized trial. In Open Forum Infectious Diseases (Vol. 5, No. 8, p. ofy182). US: Oxford University Press. https://doi.org/10.1093/ofid/ofy182
Doyle, C. J., Fitzsimmons, T. R., Marchant, C., Dharmapatni, A. A. S. S. K., Hirsch, R., & Bartold, P. M. (2015). Azithromycin suppresses P. gingivalis LPS-induced pro-inflammatory cytokine and chemokine production by human gingival fibroblasts in vitro. Clinical Oral Investigations, 19, 221-227. https://doi.org/10.1007/s00784-014-1249-7
Ellison, C. A., & Blackwell, S. B. (2021). Acute hepatocellular injury associated with azithromycin. Journal of pharmacy practice, 34(3), 493-496. https://doi.org/10.1177/0897190019894428
Farhat, Z., Hershberger, P. A., Freudenheim, J. L., Mammen, M. J., Hageman Blair, R., Aga, D. S., & Mu, L. (2021). Types of garlic and their anticancer and antioxidant activity: A review of the epidemiologic and experimental evidence. European Journal of Nutrition, 1-25. https://doi.org/10.1007/s00394-021-02482-7
Feng, Y., Guo, Q., & Shao, B. (2019). Cytotoxic comparison of macrolide antibiotics and their chlorinated disinfection byproduct mixtures. Ecotoxicology and Environmental Safety, 182, 109415. https://doi.org/10.1016/j.ecoenv.2019.109415
Li, S. Q., Wan, X. D., Zhu, S., Han, H. M., Xu, Z. S., & Lu, H. J. (2016). Establishment of a new animal model of azithromycin-induced liver injury and study the molecular pathological change during the process. Human & Experimental Toxicology, 35(5), 511-525. https://doi.org/10.1177/0960327115595684
Liu, K., Wang, G., Li, L., Chen, G., Gong, X., Zhang, Q., & Wang, H. (2020). GR-C/EBPα-IGF1 axis mediated azithromycin-induced liver developmental toxicity in fetal mice. Biochemical Pharmacology, 180, 114130. https://doi.org/10.1016/j.bcp.2020.114130
Olayinka, E. T., & Ore, A. (2014). Influence of azithromycin treatment on hepatic lipid peroxidation and antioxidant defence systems of rats. British Journal of Pharmaceutical Research, 4(2), 240-256.
Oliver, M. E., & Hinks, T. S. (2021). Azithromycin in viral infections. Reviews in Medical Virology, 31(2), e2163. https://doi.org/10.1002/rmv.2163
Omara, F., Aziz, S. A., El-Sheikh, S. M., & Said, M. A. A. (2021). Ascorbic acid attenuated the hepatic parenchymal necrosis induced by azithromycin-etoricoxib interaction in rats. Journal of Animal Health and Production, 9(1), 42-48. http://dx.doi.org/10.17582/journal.jahp/2021/9.s1.42.48
Radwan, D. A., Hussin, M. A. E., & Mahmoud, A. (2021). Antioxidant Approach of Nigella Sativa, Vit C and Silymarin for amelioration of Azithromycin induced Hepatotoxicity: Histological, Immunohistochemical and Biochemical study. Egyptian Society of Clinical Toxicology Journal, 9(2), 29-45. https://dx.doi.org/10.21608/esctj.2021.106959.1005
Sheikh Raisuddin, S. A., Fatima, M., & Dabeer, S. (2018). Toxicity of anticancer drugs and its prevention with special reference to role of garlic constituents. Annals of Phytomedicine, 7(1), 13-26.
Shukry, M., Alotaibi, S. S., Albogami, S. M., Fathallah, N., Farrag, F., Dawood, M. A., & Gewaily, M. S. (2020). Garlic alleviates the injurious impact of cyclosporine-A in male rats through modulation of fibrogenic and steroidogenic genes. Animals, 11(1), 64. https://doi.org/10.3390/ani11010064
Sinha, S., Adhikari, K., Shankarbabu, T., & Sinha, K. T. (2017). Azithromycin–the unsung hero in periodontics. IP International Journal of Periodontology and Implantology, 2(1), 1-4.
Woodhead, J. L., Yang, K., Oldach, D., MacLauchlin, C., Fernandes, P., Watkins, P. B., ... & Howell, B. A. (2019). Analyzing the mechanisms behind macrolide antibiotic-induced liver injury using quantitative systems toxicology modeling. Pharmaceutical research, 36, 1-12. https://doi.org/10.1007/s11095-019-2582-y
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Biomedicine and Chemical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.



















