Design and Synthesis Ligands Tetradents Substituted with Halogenes in α- Position and Conjugation with Riboflavin (Bioconjugates)
Conjugate ligands Type TPA’s with Flavonoids as un Electron Mediator
DOI:
https://doi.org/10.48112/bcs.v1i2.85Abstract
 Abstract Views: 346
Abstract Views: 346
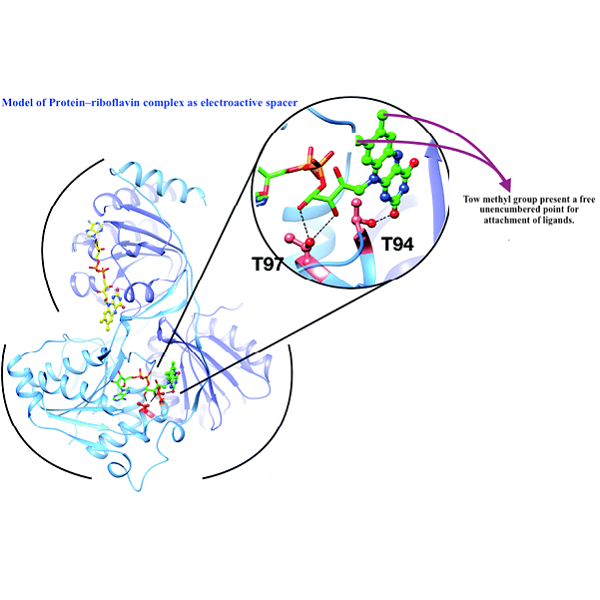
In this article, we describe the process of binding riboflavin to a simple tetradents ligand substituted in α- position from TPAs types, by reacting bromotetraacetate riboflavin with α- substituted TPA with one of the pyridine rings by nitrile group and the two other pyridine rings by halogen atoms. This type of ligands showed very important properties for the activation and transfer of oxygen to a substrate in presence of iron salt. After the tetradents were obtained, the nitrile group was reduced to an amine group where it reacts with Boc group to protect one of the amine hydrogen and then bound to the bromotetraacetate through the amine group under special reactive conditions, to form the α-8-TPAs N- Ac4riboflavin ligands. This compound can be described as a molecular tweezers in which the flavin moiety acts as a potential electron mediator.
Keywords:
TPA’s, N ligands, Riboflavin, Electron MediatorReferences
Blackman, A. G. (2005). The coordination chemistry of tripodal tetraamine ligands. Polyhedron, 24(1), 1-39. https://doi.org/10.1016/j.poly.2004.10.012
Caprio, V., & Mann, J. (1998). Synthesis of novel chromeno [3, 4-b] pyridinones. Journal of the Chemical Society, Perkin Transactions 1, (19), 3151-3156. https://doi.org/10.1039/A805650A
Chuang, C. L., Dos Santos, O., Xu, X., & Canary, J. W. (1997). Synthesis and cyclic voltammetry studies of copper complexes of Bromo-and Alkoxyphenyl-substituted derivatives of Tris (2-pyridylmethyl) amine: influence of cation− Alkoxy interactions on copper redox potentials. Inorganic chemistry, 36(9), 1967-1972. https://doi.org/10.1021/ic960942y
Harata, M., Jitsukawa, K., Masuda, H., & Einaga, H. (1995). Synthesis and structure of a new tripodal polypyridine copper (II) complex that enables to recognize a small molecule. Chemistry letters, 24(1), 61-62. https://doi.org/10.1246/cl.1995.61
Machkour, A., Mandon, D., Lachkar, M., & Welter, R. (2004). Easy preparation of the tris (2-fluoro-6-pyridylmethyl) amine ligand and instantaneous reaction of the corresponding dichloroferrous complex with molecular dioxygen: New access to dinuclear species. Inorganic chemistry, 43(4), 1545-1550. https://doi.org/10.1021/ic034485e
Machkour, A., Thallaj, N. K., Benhamou, L., Lachkar, M., & Mandon, D. (2006). The Coordination Chemistry of FeCl3 and FeCl2 to Bis[2-(2,3-dihydroxyphenyl)-6-pyridylmethyl](2-pyridylmethyl)amine: Access to a Diiron(III) Compound with an Unusual Pentagonal-Bipyramidal/Square-Pyramidal Environment. Chemistry–A European Journal, 12(25), 6660-6668. https://doi.org/10.1002/chem.200600276
Malek, Z. S., & Labban, L. M. (2021). Photoperiod regulates the daily profiles of tryptophan hydroxylase-2 gene expression the raphe nuclei of rats. International Journal of Neuroscience, 131(12), 1155-1161. https://doi.org/10.1080/00207454.2020.1782903
Malek, Z. S., Dardente, H., Pevet, P., & Raison, S. (2005). Tissue‐specific expression of tryptophan hydroxylase mRNAs in the rat midbrain: anatomical evidence and daily profiles. European Journal of Neuroscience, 22(4), 895-901. https://doi.org/10.1111/j.1460-9568.2005.04264.x
Malek, Z. S., Dardente, H., Pevet, P., & Raison, S. (2005). Tissue‐specific expression of tryptophan hydroxylase mRNAs in the rat midbrain: anatomical evidence and daily profiles. European Journal of Neuroscience, 22(4), 895-901. https://doi.org/10.1111/j.1460-9568.2005.04264.x
Malek, Z. S., Pevet, P., & Raison, S. (2004). Circadian change in tryptophan hydroxylase protein levels within the rat intergeniculate leaflets and raphe nuclei. Neuroscience, 125(3), 749-758. https://doi.org/10.1016/j.neuroscience.2004.01.031
Malek, Z. S., Pevet, P., & Raison, S. (2004). Circadian change in tryptophan hydroxylase protein levels within the rat intergeniculate leaflets and raphe nuclei. Neuroscience, 125(3), 749-758. https://doi.org/10.1016/j.neuroscience.2004.01.031
Malek, Z. S., Sage, D., Pévet, P., & Raison, S. (2007). Daily rhythm of tryptophan hydroxylase-2 messenger ribonucleic acid within raphe neurons is induced by corticoid daily surge and modulated by enhanced locomotor activity. Endocrinology, 148(11), 5165-5172. https://doi.org/10.1210/en.2007-0526
Malek, Z. S., Sage, D., Pévet, P., & Raison, S. (2007). Daily rhythm of tryptophan hydroxylase-2 messenger ribonucleic acid within raphe neurons is induced by corticoid daily surge and modulated by enhanced locomotor activity. Endocrinology, 148(11), 5165-5172. https://doi.org/10.1210/en.2007-0526
Malek, Z., & Labban, L. (2019). A comparative study of tryptophan hydroxylase's circadian rhythm in the functional parts of dorsal raphe nuclei in the mesencephalon. European Journal of Pharmaceutical and Medical Research, 6(11), 527-532.
Mandon, D., Machkour, A., Goetz, S., & Welter, R. (2002). Trigonal bipyramidal geometry and tridentate coordination mode of the tripod in FeCl2 complexes with tris (2-pyridylmethyl) amine derivatives bis-α-substituted with bulky groups. Structures and spectroscopic comparative studies. Inorganic chemistry, 41(21), 5364-5372. https://doi.org/10.1021/ic011104t
Miyaura, N., Yanagi, T., & Suzuki, A. J. S. C. (1981). The palladium-catalyzed cross-coupling reaction of phenylboronic acid with haloarenes in the presence of bases. Synthetic Communications, 11(7), 513-519. https://doi.org/10.1080/00397918108063618
Murray, T. A., & Swenson, R. P. (2003). Mechanism of flavin mononucleotide cofactor binding to the Desulfovibrio vulgaris flavodoxin. 1. Kinetic evidence for cooperative effects associated with the binding of inorganic phosphate and the 5 ‘-phosphate moiety of the cofactor. Biochemistry, 42(8), 2307-2316. https://doi.org/10.1021/bi026967s
Murray, T. A., Foster, M. P., & Swenson, R. P. (2003). Mechanism of flavin mononucleotide cofactor binding to the Desulfovibrio vulgaris flavodoxin. 2. Evidence for cooperative conformational changes involving tryptophan 60 in the interaction between the phosphate-and ring-binding subsites. Biochemistry, 42(8), 2317-2327. https://doi.org/10.1021/bi026968k
Offermann, W., & Vögtle, F. (1980). Brominations with N‐Bromosuccinimide: Solvent and Selectivity. Angewandte Chemie International Edition in English, 19(6), 464-465. https://doi.org/10.1002/anie.198004641
Prévot-Halter, I., Smith, T. J., & Weiss, J. (1997). Assembling organic receptors around transition metal templates: functionalized catechols and dioxomolybdenum (VI) for the recognition of dicarboxylic acids. The Journal of Organic Chemistry, 62(7), 2186-2192. https://doi.org/10.1021/jo960836d
Pueyo, J. J., Curley, P. G., & Mayhew, S. G. (1996). Kinetics and thermodynamics of the binding of riboflavin, riboflavin 5′-phosphate and riboflavin 3′, 5′-bisphosphate by apoflavodoxins. Biochemical journal, 313(3), 855-861. https://doi.org/10.1042/bj3130855
Romary, J. K., Zachariasen, R. D., Barger, J. D., & Schiesser, H. (1968). New 2-pyridyl polyamines. Synthesis, spectra, and proton dissociation constants. Journal of the Chemical Society C: Organic, 2884-2887. https://doi.org/10.1039/J39680002884
Shuman, R. T., Ornstein, P. L., Paschal, J. W., & Gesellchen, P. D. (1990). An improved synthesis of homoproline and derivatives. The Journal of Organic Chemistry, 55(2), 738-741. https://doi.org/10.1021/jo00289a058
Thallaj, N. (2021). Synthesis of a New Ligand Tris (2-pyridylmethyl) amine functionalized by a methoxy group and study of Dichloroferrous complexes, its reactivity to dioxygen both in the presence and absence of substrate. International Journal of Applied Chemical and Biological Sciences, 2(4), 65-77.
Thallaj, N. (2021). Synthesis of a New Ligand Tris (2-pyridylmethyl) amine functionalized by a methoxy group and study of Dichloroferrous complexes, its reactivity to dioxygen both in the presence and absence of substrate. International Journal of Applied Chemical and Biological Sciences, 2(4), 65-77. https://identifier.visnav.in/1.0001/ijacbs-21f-07003/
Thallaj, N. K., Machkour, A., Mandon, D., & Welter, R. (2005). Square pyramidal geometry around the metal and tridentate coordination mode of the tripod in the [6-(3′-cyanophenyl)-2-pyridylmethyl] bis (2-pyridylmethyl) amine FeCl 2 complex: a solid state effect. New Journal of Chemistry, 29(12), 1555-1558. https://doi.org/10.1039/B512108F
Thallaj, N. K., Machkour, A., Mandon, D., & Welter, R. (2005). Square pyramidal geometry around the metal and tridentate coordination mode of the tripod in the [6-(3′-cyanophenyl)-2-pyridylmethyl] bis (2-pyridylmethyl) amine FeCl 2 complex: a solid state effect. New Journal of Chemistry, 29(12), 1555-1558. https://doi.org/10.1039/B512108F
Thallaj, N. K., Mandon, D., & White, K. A. (2007). The design of metal chelates with a biologically related redox-active part: Conjugation of riboflavin to bis (2-pyridylmethyl) amine ligand and preparation of a ferric complex. European journal of inorganic chemistry, (1), 44-47. https://doi.org/10.1002/ejic.200600789
Thallaj, N. K., Mandon, D., & White, K. A. (2007).The design of metal chelates with a biologically related redox-active part: Conjugation of riboflavin to bis (2-pyridylmethyl) amine ligand and preparation of a ferric complex. European journal of inorganic chemistry, (1), 44-47. https://doi.org/10.1002/ejic.200600789
Thallaj, N. K., Orain, P. Y., Thibon, A., Sandroni, M., Welter, R., & Mandon, D. (2014). Steric Congestion at, and Proximity to, a Ferrous Center Leads to Hydration of α-Nitrile Substituents Forming Coordinated Carboxamides. Inorganic chemistry, 53(15), 7824-7836. https://doi.org/10.1021/ic500096h
Thallaj, N. K., Przybilla, J., Welter, R., & Mandon, D. (2008). A ferrous center as reaction site for hydration of a nitrile group into a carboxamide in mild conditions. Journal of the American Chemical Society, 130(8), 2414-2415. https://doi.org/10.1021/ja710560g
Thallaj, N. K., Przybilla, J., Welter, R., & Mandon, D. (2008). A ferrous center as reaction site for hydration of a nitrile group into a carboxamide in mild conditions. Journal of the American Chemical Society, 130(8), 2414-2415. https://doi.org/10.1021/ja710560g
Tyeklar, Z., Jacobson, R. R., Wei, N., Murthy, N. N., Zubieta, J., & Karlin, K. D. (1993). Reversible reaction of dioxygen (and carbon monoxide) with a copper (I) complex. X-ray structures of relevant mononuclear Cu (I) precursor adducts and the trans-(. mu.-1, 2-peroxo) dicopper (II) product. Journal of the American Chemical Society, 115(7), 2677-2689. https://doi.org/10.1021/ja00060a017
Walker, W. H., Singer, T. P., Ghisla, S., & Hemmerich, P. (1972). Studies on Succinate Dehydrogenase: 8α‐Histidyl‐FAD as the Active Center of Succinate Dehydrogenase. European Journal of Biochemistry, 26(2), 279-289. https://doi.org/10.1111/j.1432-1033.1972.tb01766.x
Walsh, M. A., McCarthy, A., O'Farrell, P. A., McArdle, P., Cunningham, P. D., Mayhew, S. G., & Higgins, T. M. (1998). X‐ray crystal structure of the Desulfovibrio vulgaris (Hildenborough) apoflavodoxin‐riboflavin complex. European journal of biochemistry, 258(2), 362-371. https://doi.org/10.1046/j.1432-1327.1998.2580362.x
Wane, A., Thallaj, N. K., & Mandon, D. (2009). Biomimetic Interaction between FeII and O2: Effect of the Second Coordination Sphere on O2 Binding to FeII Complexes: Evidence of Coordination at the Metal Centre by a Dissociative Mechanism in the Formation of μ‐Oxo Diferric Complexes. Chemistry–A European Journal, 15(40), 10593-10602. https://doi.org/10.1002/chem.200901350
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Biomedicine and Chemical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.



















